Electrodeposition Blog Posts

Optimizing the Electroplating Process for Multiple Components
From everyday kitchen utensils to iconic award show statuettes, rack plating is a useful manufacturing process that ensures components are electroplated both uniformly and efficiently.
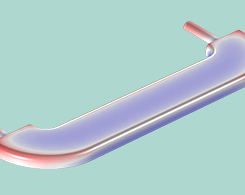
The Art and Science of Electrochemical Plating
Electrochemical plating, a surface finishing technique used in the automotive, electronics, corrosion protection, aerospace, and defense industries, is both an art and a science. Let us explain.
Modeling Copper Electrodeposition on a Circuit Board
Copper electrodeposition is used to manufacture PCBs. Here, we discuss modeling the electrodeposition of copper in the trench of a PCB.
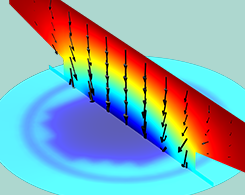
How to Model Electrochemical Resistance and Capacitance
Get an overview of the theory and modeling considerations for resistive and capacitive effects, which are fundamental to the understanding of electrochemical systems.
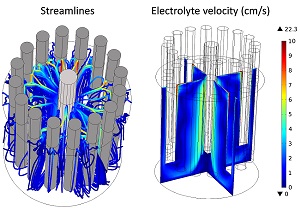
Modeling Current Distributions in a Molten Salt Electro-Refiner
A guest blogger from SIMTEC discusses how he uses simulation to analyze current distributions in a molten salt electrorefiner. Get details here >>
Electroplating Simulations for Printed Circuit Board Designers
The manufacture of a printed circuit board (PCB) often involves a process called electroplating, which can vary from design to design. A simulation app can make it easy to analyze this process.
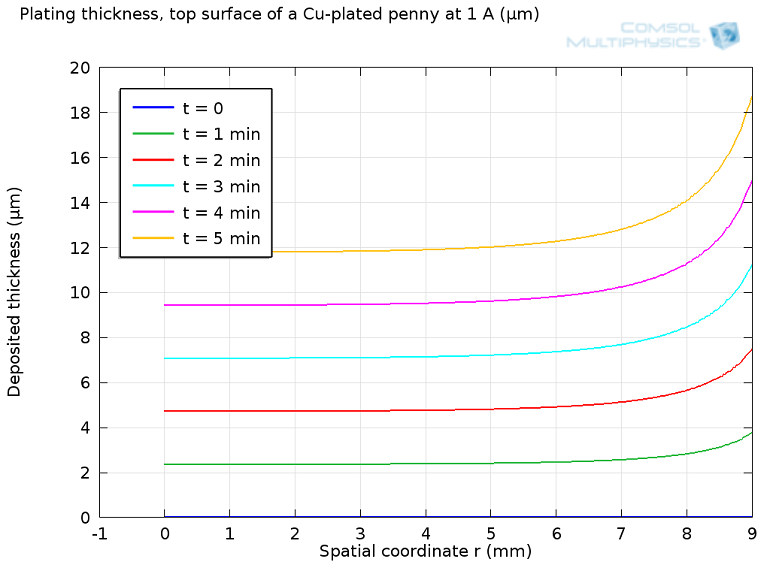
Electroplating: How the U.S. Mint Makes a Penny
Did you know that a penny actually doesn’t contain a lot of copper? Learn about how the U.S. Mint makes pennies through the process of electroplating, which can be studied with chemical modeling.
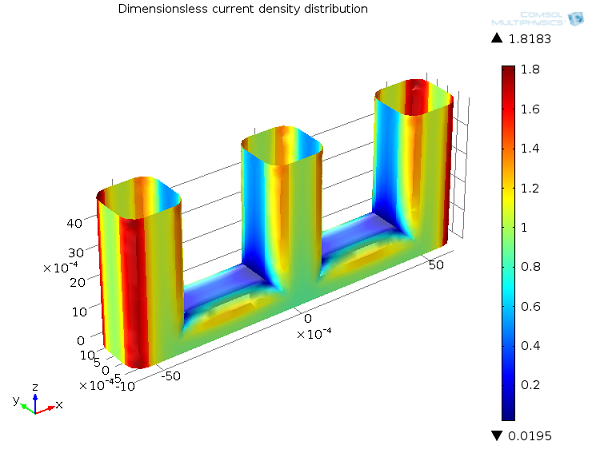
Which Current Distribution Interface Do I Use?
See the different current distributions with a wire electrode example to help you choose between the current distribution interfaces in COMSOL Multiphysics® for your electrochemical simulations.
